Autologous intraoperative blood salvage (IBS) is used routinely in many surgical specialties to minimize the effects of intraoperative bleeding, avoiding the risks of allogeneic red blood cell (RBC) transfusion. A recent cochrane review showed a 40% reduction in the requirements for allogeneic blood transfusion with cell salvage[1]. IBS has been generally used in liver transplantation (LT), although it is not usually recommended in patients with hepatocellular carcinoma (HCC) since there is a putative risk of reinfusion of neoplastic cells. The IBS is an alternative to allogeneic blood transfusion but it remains a controversial technique in oncologic procedures since it could represent an uncertain risk of malignant cell reinfusion[2–5].
The circulation of viable neoplastic cells in the IBS device and their detection in the leukocyte depletion filter (LDF) have been proved, and LDF has been used as an effective method to clean the RBC component before infusing it back[5–9]. Although the rationale to use LDF to block neoplastic cells back by the IBS device has been investigated on experimental studies, the clinical relevance analysis over patients who underwent LT for HCC has been restricted to a single study[5]. In the latter case, no differences were observed in recurrence between patients who received IBS and those who did not. However, it was not possible to rule out the possibility that this result was a consequence of a small sample size.
The aim of this study was to evaluate if the use of IBS for HCC patients who underwent LT increases the risk of tumor recurrence. To our knowledge, this is the largest series addressing this question in this population.
Patients submitted to LT for HCC at Hospital das Clínicas of University of São Paulo Medical School (HCFMUSP) were analyzed from a prospectively maintained database containing demographic, clinical, operative, pathological, and follow-up data and studied retrospectively. Permission was obtained from the informed consent statement and institutional review board according to the institutional policy for protected health information.
All patients presented in this analysis were initially considered to meet the Milan criteria or UCSF criteria[10,11]. Patients who had detectable extra-hepatic disease during the pre- or intraoperative course and patients with a concurrent second neoplasm were not included. Patients who did not present HCC in the specimen were excluded with the exception of those previously treated with radiofrequency or chemoembolization. Pre-operative imaging modalities to evaluate the extent of intrahepatic disease and to exclude extra-hepatic metastatic sites included computed tomography and/or magnetic resonance imaging of the chest, abdomen, and pelvis. Model of end-stage liver disease (MELD) scores were calculated using laboratory results collected prior to the LT. The MELD score was calculated using the standard UNOS formula: MELD = 3.78 × ln (bilirubin) + 11.2 × ln (INR) + 9.57 × ln (creatinine) + 6.43, where bilirubin and creatinine are in mg/dL units and INR is the international normalized ratio. The MELD score was analyzed separately as both continuous and categorical variables (i.e., ≥ 20 vs <20).
The estimated blood loss was not fully available and thus it was not described and analyzed. The intraoperative decision to transfuse either allogeneic or autologous blood was consensual between the surgeon and the anesthesiologist. It was based on hemodynamic status, blood loss, hemoglobin concentration and patient’s comorbidities.
Follow-up time was calculated from the date of LT to the date of last clinical encounter captured by the HCFMUSP medical record system or the date of death. Recurrence-free survival (RFS) was calculated from the LT to the first detected recurrence or last follow-up without recurrence. Overall survival (OS) was calculated based on the survivorship status (deceased or alive) at last follow-up.
The blood from the surgical field was collected using a Cell Saver auto-transfusion device (Fresenius C.A.T.S, Terumo Cardiovascular Systems, Germany) and anti-coagulated with heparinized saline and stored. The RBC component of aspirated blood was centrifuged and washed with heparinized saline. The RBC concentrates were filtered through an LDF (FTS-RC202, Shuangweibio Corp., Nanjing, China). Processed RBCs were transfused back to the patient when appropriate.
Statistical analyses were performed using the Fisher’s exact test and the Wilcoxon rank sum test to examine covariate differences between patients who underwent IBS and those who did not. Values are expressed as median (interquartile) or percentage, as appropriate. Survival probabilities were estimated using the Kaplan-Meier method and compared using the Log-Rank test. A Cox regression model was developed to determine factors independently associated with death. The use of IBS was included in the multivariate analysis regardless of its univariate significance. Other factors that were significantly associated with outcomes by univariate analysis (inclusion criterion, P≤ 0.1) were entered into a multivariate analysis to test for significance of IBS adjusting for possible confounders. For recurrence assessment, no Cox regression was used since the number of events per variable was not appropriated[12,13]. A P value < 0.05 was considered significant for univariate and multivariate analyses. All statistical analyses were conducted using STATA v 9.0 (Stata Corp, College Station, TX).
Between January 2002 and September 2012, 158 consecutive patients who underwent potentially curative LT for HCC were included. One hundred and twenty-two (77.2%) patients in the IBS group and 36 (22.8%) patients in the non-IBS group were compared. Patients and clinicopathological presentation were compared between groups and are summarized in Table 1. Briefly, the demographic and clinicopathological characteristics were comparable between the two groups. The only significant difference was the presence of liver cirrhosis, which was more prevalent in the non-IBS group (100% vs 84.8%, P = 0.014).
| Total (%) n = 158 | Intraoperative blood salvage
|
P | ||
| Yes (%) n = 122 (77.2) | No (%) n = 36 (22.8) | |||
| Age1 | 58 (51-62) | 58 (51-62) | 58 (51-62) | 0.958 |
| Male gender | 122 (77.2) | 95 (77.9) | 27 (75) | 0.821 |
| BMI12 | 25.7 (23.6-27.8) | 25.7 (23.6-27.8) | 25.5 (23.5-2.3) | 0.712 |
| Pre-op AFP3 | 9.2 (3.7-35.4) | 8.9 (3.5-3.6) | 10.9 (6.7-33.7) | 0.175 |
| Cirrhosis4 | 135 (88.3) | 100 (84.8) | 35 (100) | 0.014 |
| Alcohol4 | 22 (14.4) | 18 (15.3) | 4 (11.4) | 0.785 |
| Hepatitis4 | ||||
| B | 20 (13.1) | 12 (10.2) | 8 (22.9) | 0.082 |
| C | 97 (63.4) | 73 (61.9) | 24 (68.6) | 0.551 |
| Others4 | 8 (5.2) | 8 (6.8) | 0 | 0.199 |
| Blood type | 0.420 | |||
| A | 60 (37) | 42 (34.4) | 18 (50) | |
| B | 21 (13.3) | 17 (13.9) | 4 (11.1) | |
| AB | 14 (8.9) | 11 (9) | 3 (8.3) | |
| O | 63 (39.9) | 52 (42.6) | 11 (30.6) | |
| Rhesus5 | 123 (86.6) | 93 (86.1) | 30 (88.3) | 1.000 |
| MELD1 | 10 ( 8-15) | 10.5 (9-17) | 9 (8-13.5) | 0.058 |
| Radiofrequency4 | 4 (2.6) | 3 (2.6) | 1 (2.8) | 1.000 |
| Chemoembolization4 | 69 (45.1) | 53 (45.3) | 16 (44.5) | 1.000 |
| Alcoholization4 | 7 (4.6) | 5 (4.3) | 2 (5.6) | 0.668 |
| Graft/body proportion12 | 1.75 (1.5-2.2) | 1.8 (1.5-2.2) | 1.7 (1.4– 2.2) | 0.454 |
| Number of lesions1 | 2 (1-3) | 2 (1-3) | 2 (1-3) | 0.715 |
| Largest lesion, mm1 | 25 (19-31) | 25 (19-30) | 25 (18-35) | 0.384 |
| Edmond-steiner degree (III and IV)6 | 88 (59.9) | 67 (58.8) | 21 (63.5) | 0.689 |
| Vascular invasion | 53 (33.6) | 44 (36.1) | 9 (25) | 0.236 |
| Microsatellite lesions | 26 (16.5) | 19 (15.6) | 7 (19.4) | 0.612 |
| Cholangiocarcinoma | 6 (3.8) | 6 (4.9) | 0 | 0.338 |
| Recurrence | 14 (8.9) | 10 (8.2) | 4 (11.1) | 0.525 |
| Death | 52 (32.9) | 41 (33.6) | 11 (30.6) | 0.841 |
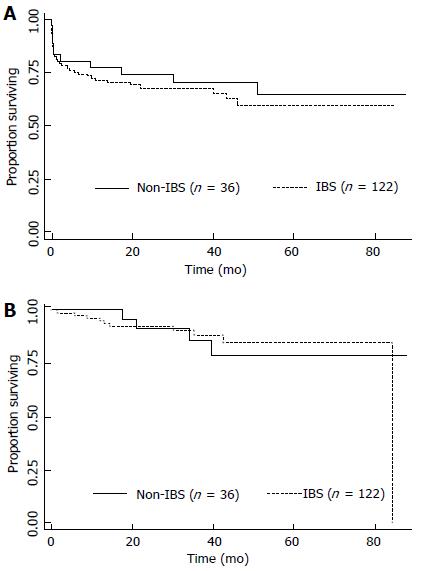
The median follow-up time for all patients was 27 mo; 25 mo for the group who received IBS and 32 mo for the group who did not (P = 0.049). The median follow-up time for survivors was 38 mo; 37 mo for the group who received IBS and recurred and 41 mo for the group who did not (P = 0.017). The estimated 3- and 5-year OS rates were 68% and 59.7%, respectively. When OS was adjusted for the use of IBS or not, no difference was detected (P = 0.51), as depicted in the Figure 1A. The univariate and multivariate analyses for death were performed and are shown in Table 2. Briefly, no differences were detected according to MELD either as a continuous variable (recurrence, P = 0.633; death, P = 0.286) or as binominal, as demonstrated in Tables 2 and 3. Only elevated Edmond-Steiner degree of tumor differentiation (III-IV) remained significant for the risk of death, as shown in Table 2. The estimated 3- and 5-year RFS rates were 87.7% and 83.3%, respectively. When RFS was adjusted for the use of IBS or not, no difference was detected (P = 0.953; Figure 1B). The univariate analysis for recurrence is shown in Table 3. Briefly, elevated Edmond-Steiner degree of tumor differentiation (III-IV), pre-operative alpha-feto protein level equal to or higher than 100 ng/dL and presence of microsatellite lesions were independent predictors of recurrence, as demonstrated in Table 3.
| Total | 5-yr survival (%) | Median survival (mo) | Univariate analysis P | HR | 95%CI | Multivariate analysis P | |
| Overall | 158 | 59.7 | – | – | |||
| Age (≥ 60 yr) | – | – | – | 0.133 | |||
| Gender | – | – | 0.097 | ||||
| Male | 122 | 61.5 | 0.88 | 0.45-1.74 | 0.714 | ||
| Female | 36 | 55.4 | – | ||||
| BMI (≥ 28) | 0.080 | ||||||
| Yes | 37 | 48.2 | 46 | 1.55 | 0.81-2.98 | 0.186 | |
| No | 113 | 63.6 | – | ||||
| Pre-op AFP (≥ 100 ng/dL) | 0.087 | ||||||
| Yes | 19 | 51.8 | – | 1.50 | 0.68-3.32 | 0.316 | |
| No | 129 | 60.8 | – | ||||
| Cirrhosis | – | – | – | 0.950 | |||
| Alcohol related | 0.048 | ||||||
| Yes | 22 | 86.4 | – | 0.30 | 0.09-1 | 0.051 | |
| No | 131 | 55.5 | – | ||||
| Hepatitis B infection | – | – | – | 0.156 | |||
| Hepatitis C infection | – | – | – | 0.130 | |||
| Others | – | – | – | 0.281 | |||
| Blood type | – | – | – | 0.470 | |||
| Rhesus | – | – | – | 0.554 | |||
| Radiofrequency | – | – | – | 0.821 | |||
| MELD (≥ 15) | – | – | – | 0.721 | |||
| Chemo-embolization | – | – | – | 0.877 | |||
| Tumor alcoholization | – | – | – | 0.118 | |||
| Graft/body % (≥ 2) | – | – | – | 0.163 | |||
| No. of lesions (> 3) | – | – | – | 0.819 | |||
| Largest lesion (≥ 30 mm) | – | – | – | 0.640 | |||
| Edmond-Steiner degree | 0.013 | ||||||
| III-IV | 88 | 48.9 | 51 | 2.19 | 1.07-4.47 | 0.031 | |
| 0-II | 59 | 74.4 | – | ||||
| Vascular invasion | – | – | – | 0.290 | |||
| Microsatellite lesions | – | – | – | 0.283 | |||
| Cholangiocarcinoma | – | – | – | 0.957 | |||
| IBS | 0.510 | ||||||
| Yes | 122 | 59.5 | – | 1.56 | 0.74-3.30 | 0.237 | |
| No | 36 | 64.5 | – | ||||
| Total | 5-yr survival (%) | Median survival (mo) | Univariate analysis P | |
| Overall | 158 | 83.3 | – | – |
| Age (≥ 60 yr) | – | – | – | 0.319 |
| Male gender | – | – | – | 0.410 |
| BMI (≥ 28) | – | – | – | 0.166 |
| Pre-op AFP (≥ 100 mg/dL) | 0.001 | |||
| Yes | 19 | 59.4 | 84.5 | |
| No | 129 | 85 | – | |
| Cirrhosis | – | – | – | 0.163 |
| Alcohol related | – | – | – | 0.207 |
| Hepatitis B infection | – | – | – | 0.911 |
| Hepatitis C infection | – | – | – | 0.568 |
| Others | – | – | – | 0.794 |
| Blood type | – | – | – | 0.912 |
| Rhesus | – | – | – | 0.494 |
| MELD (≥ 15) | – | – | – | 0.694 |
| Radiofrequency | – | – | – | 0.758 |
| Chemoembolization | – | – | – | 0.133 |
| Tumor alcoholization | – | – | – | 0.373 |
| Graft/body % (≥ 2) | – | – | – | 0.605 |
| Number of lesions (> 3) | – | – | – | 0.496 |
| Largest lesion mm (≥ 30) | – | – | – | 0.429 |
| Edmond-Steiner degree | 0.0162 | |||
| III-IV | 88 | 73 | 84.5 | |
| 0-II | 59 | 94.3 | – | |
| Vascular invasion | 0.071 | |||
| Yes | 26 | 74.8 | 84.5 | |
| No | 132 | 86.3 | – | |
| Microsatellite lesions | 0.007 | |||
| Yes | 26 | – | – | |
| No | 132 | 86.5 | – | |
| Cholangiocarcinoma | – | – | – | 0.375 |
| IBS | 0.953 | |||
| Yes | 122 | 85 | 84.5 | |
| No | 36 | 78.8 | – |
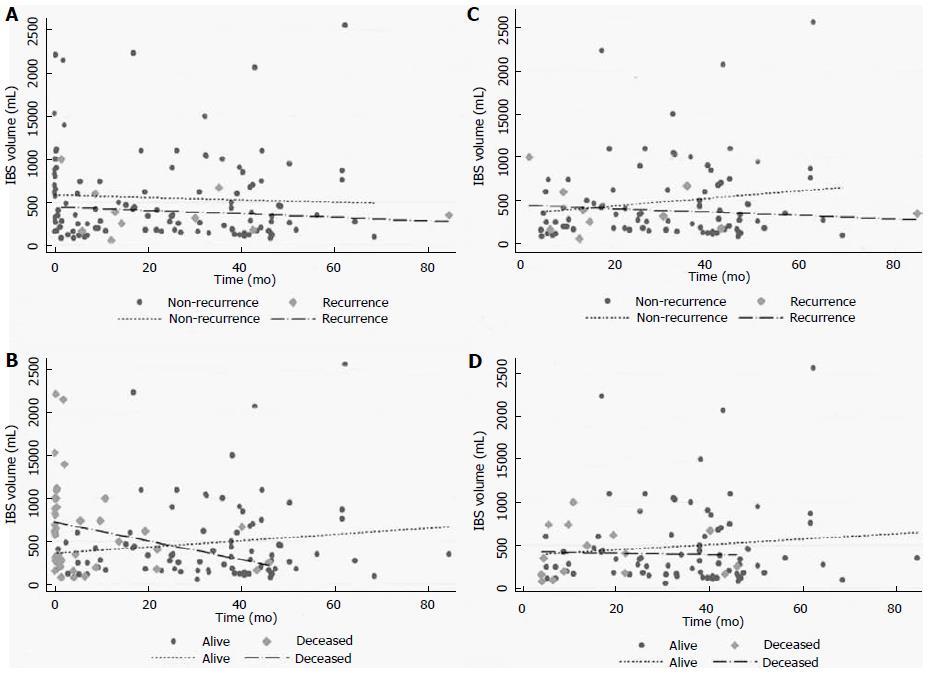
Regarding the group of patients who received IBS (122 patients), the infusion volume was additionally analyzed as a continuous variable, and no differences were found in either recurrence (P = 0.512) or death (P = 0.055), as demonstrated in Figure 2A and B. Analyses of outcomes at 90 d or longer were performed and no differences in recurrence (P = 0.518) or death (P = 0.518) were detected (Figure 2C and D).
The IBS is largely accepted as an option for blood transfusion. However, the contra-indications are based on the use of contaminated blood as in chronic diseases like hepatitis or other viral infections, bile infection or colonization, and intra-operative contamination[4,8]. The same rationale is applied to avoid tumor dissemination in patients with liver cancer already identified. Although this apprehension has been justifying its practice, no clear relation between the use of IBS and cancer recurrence has already been proved. Operations with high blood loss including cancer surgery have been demanding IBS use, however retrospective series did not show any suggestive association between the increase of recurrence and IBS use[14].
Concerning HCC patients, IBS use was described in a few series for resection and LT. One series described no increase in recurrence with IBS, showing no differences in higher stages and even better results for patients who used IBS in early stage disease[15,16]. Two series of LT, respectively, with 31 and 40 patients in the IBS groups vs 16 and 96 patients as control group, were described[17,18]. Despite the theoretical risk of tumor cell dissemination, the recurrence rates were not increased by IBS use in both series[17,18].
The purpose of our study was to compare long-term outcomes for patients undergoing LT for HCC who received IBS or not. In our study population, the groups were comparable except for the remarkable presence of cirrhosis in the IBS group. As expected, patients with cirrhosis are technically challenging and the blood loss is usually elevated, more justifying IBS. With regard to oncologic outcomes, the use of IBS or not was not significantly associated with recurrence or death. The predictors associated with recurrence were presence of satellite lesions and elevated Edmond-Steiner tumor degree. This was also an independent predictor of death in the multivariate model. The principal finding of this study is that in a large patient population from a single institution there were no measurable differences in outcomes based on the IBS use for patients who underwent LT for HCC.
Regarding only the IBS group, differences in the volumes infused were associated with death but not with recurrence, as depicted in Figure 2. The volume infused changed when the time point of 90 d was used. In the earlier period, higher volumes were associated with death but not with recurrence. This performance translates the IBS volume as surrogate of estimated blood loss, which is an independent predictor of mortality and transfusion as well[19]. Patients in the earlier period died in a short follow-up period and they could not have presented recurrence. With regard to longer follow-up (90 d or longer), the IBS volumes fit similarly for the distribution of recurrence or death. Long-term outcomes were not affected for the IBS volume in our series.
The limitations of the study are those associated with the immeasurable biases seen in all retrospective studies. We recognize that selection bias based on several nonobjective, undocumented criteria may have contributed to some of the differences between the two study groups. The estimated blood loss was not fully available and thus it was not described or analyzed.
The major finding of this analysis is the lack of any association between the use of IBS and oncologic outcomes. The results of this study should not be misinterpreted as an endorsement for the IBS use for all cancer patients. On the contrary, our data claim for more translational and clinical investigations of this issue. The operative hemorrhage in LT remains significant and blood transfusion is often demanded. The IBS should be applied as much as necessary, however the rationale of tumor cell reinfusion is a common concern[4,14,17,18,20–22]. Studies in vitro and retrospective series suggest that the use of LDF is effective enough to avoid tumor cell recirculation[5–7]. We believe that this finding is convincing and perhaps it is a reasonable explanation for no differences in recurrence or death in our series, since the LDF was used in all cases.
Moreover, a recent meta-analysis, including only non-randomized trials, showed an increase of risk for death and recurrence in patients with HCC who received allogeneic blood transfusion during hepatic resection[23]. Patients in the allogeneic group had a 16% more chance of recurrence at 5 years as well as a 60% more chance of all-case death in the same period. The reasons for the worse outcomes remain uncertain but it has been assumed that suppressive effects in the host immune system may have been responsible. The postulated mechanisms are allogeneic mononuclear cells; leucocytes-derived soluble mediators; and soluble HLA peptides circulating in allogeneic plasma inducing the host immune suppression[24]. These effects could be prevented by the autologous transfusion[24].
In summary, the present study shows that in a large consecutive series of patients undergoing LT for HCC in this single institution, there were no measurable differences in RFS or OS between patients who received IBS or not. With the lack of randomized clinical trials comparing the use of IBS for oncologic patients, its use could be considered a reasonable option for individualized patients. Based on these data, a trial looking for no inferiority comparing the use of IBS and conventional blood transfusion for LT for HCC is justified and should be designed to detect small differences in outcomes.
Blood transfusion is usually necessary for liver transplantation (LT). Intra-operative blood salvage has generally been used in LT to avoid deleterious effect of allogeneic blood transfusion. However, autologous blood transfusion has not been recommended in patients with hepatocellular carcinoma (HCC) since there is a putative risk of reinfusion of neoplastic cells.
Although there is a putative risk of reinfusion of cancer cells into circulation during surgery, there is no data yet demonstrating that it would really impact on oncologic outcomes. This study did not demonstrate impact on clinical and oncologic outcomes. However, since the data are retrospective, our finding claims for trials looking for no inferiority comparing the two modalities of blood transfusion in patients who underwent LT for HCC, to detect small differences in outcomes.
This study addresses an alternative option for allogeneic blood transfusion during LT for HCC. The autologous blood salvage in LT, in this series, did not impact recurrence or death. This suggests that autologous blood transfusion should be considered an option avoiding the deleterious effects of allogeneic blood transfusion.
The use of intra-operative blood salvage would have immunological and economic impact during postoperative course. Circulating cancer cells were already demonstrated, however it also seems that leucocyte filters are safe enough to block those cells. Then, the use of auto-transfusion devices associated with leucocytes filters seems to be a potential resource to help patients who undergo LT for HCC
IBS: Autologous intraoperative blood salvage; HCC: Hepatocellular carcinoma; LDF: Leukocyte depletion filter; LT: Liver transplantation; MELD: Model of End-Stage Liver Disease; OS: Overall survival; RFS: Recurrence free survival; RBC: Red blood cell.
Autologous IBS is generally used in liver transplantation to minimize the effect of intraoperative bleeding. However, the peripheral blood of HCC patients may be contaminated with cancer cells or cancer-inducing virus, which can lead to potential risks of recurrence. In this study, authors investigated the association between the intraoperative use of IBS and survival of HCC patients. According to the data of a postoperative follow-up cohort, they reported that the use of IBS cannot influence the survival of HCC patients. This is an interesting study and is useful for clinicians.















In view of a number of study limitations, evaluation of ivermectin’s anti-Zika virus activity in other animal fashions and
against different Zika virus strains can be fascinating.
Hey there, You’ve done a fantastic job. I will definitely digg it and
personally suggest to my friends. I am sure they will be benefited from this website.
Hello there, You have done a great job. I’ll definitely digg it and personally suggest to my friends.
I am confident they will be benefited from
this site.
Unquestionably believe that which you stated. Your favorite justification appeared to be on the web the simplest thing
to be aware of. I say to you, I certainly get irked while people consider worries that they just do not
know about. You managed to hit the nail upon the top and defined out the whole thing without having side-effects , people could take a signal.
Will likely be back to get more. Thanks
Hello! This is my first comment here so I just wanted to give a quick shout out and say I
genuinely enjoy reading through your articles. Can you recommend any
other blogs/websites/forums that go over the same topics?
Appreciate it!
Everything is very open with a clear explanation of the issues.
It was definitely informative. Your website is extremely helpful.
Thank you for sharing!
Excellent pieces. Keep posting such kind of information on your page.
Im really impressed by it.
Hey there, You have performed an excellent job. I’ll certainly digg it and in my view
recommend to my friends. I’m sure they will be benefited from this website.
My family every time say that I am killing my time here at web, except
I know I am getting experience every day by reading thes pleasant
content.
Does your site have a contact page? I’m having trouble locating it but, I’d like to send you an email.
I’ve got some recommendations for your blog you might be interested in hearing.
Either way, great blog and I look forward to seeing it develop over time.
I want to to thank you for this good read!! I certainly
loved every little bit of it. I’ve got you book marked to check out new stuff you
post…
Also visit my page :: https://usascripthelpers.com/
Appreciate the recommendation. Let me try it out.
buy viagra online
The other day, while I was at work, my sister stole my apple ipad and tested to see if it can survive a 30 foot drop, just so she can be a youtube sensation. My iPad is now destroyed and she has 83 views.
I know this is completely off topic but I had to share
it with someone!
Online poker
payday loan
I every time used to study article in news papers but now as I am a user of net therefore from now I
am using net for articles, thanks to web.
Here is my webpage; календарь 2024 года
You’ve made some decent points there. I checked on the web
for more info about the issue and found most individuals will go
along with your views on this site.
hi!,I like your writing so so much! proportion we communicate extra approximately your post on AOL?
I require an expert in this space to solve my problem.
May be that is you! Looking forward to look you.
Aw, this was an exceptionally good post. Taking a few minutes and actual effort to generate a top notch article… but what can I say… I procrastinate
a lot and don’t seem to get anything done.
Hey there! I know this is kinda off topic nevertheless I’d figured I’d ask.
Would you be interested in trading links or maybe
guest authoring a blog post or vice-versa? My blog addresses a lot of the same topics
as yours and I think we could greatly benefit from each
other. If you might be interested feel free to shoot me an e-mail.
I look forward to hearing from you! Excellent blog by the way!
After I originally commented I seem to have clicked on the -Notify me when new comments
are added- checkbox and now each time a comment is added I recieve 4 emails with the exact same
comment. There has to be a means you can remove me from that service?
Thanks a lot!
Its not my first time to pay a visit this web site, i am visiting this
web page dailly and obtain fastidious data from here every day.
Hello i am kavin, its my first occasion to commenting anyplace, when i read this piece of writing
i thought i could also create comment due to this good post.
Someone necessarily assist to make severely posts I’d state.
This is the very first time I frequented your website page and to this point?
I amazed with the research you made to create this actual
put up extraordinary. Magnificent task!
Fantastic website. A lot of helpful info here. I am sending it to a few pals ans also sharing in delicious.
And naturally, thank you for your effort!
The simplest anxiety and depression remedy may very well be
a subjective problem and needs to be tailor-made to each individual.
Yesterday, while I was at work, my sister stole my iphone and tested to see if it can survive a 30 foot drop, just so she
can be a youtube sensation. My iPad is now
broken and she has 83 views. I know this is totally off
topic but I had to share it with someone!
Yes! Finally someone writes about cbrbet slot login.
What’s up colleagues, fastidious post and fastidious
urging commented at this place, I am really enjoying by these.
payday loan
buy viagra online
buy viagra online
watch porn video
payday loan
penis enlargement
Hey there! Would you mind if I share your blog with
my zynga group? There’s a lot of people that I think
would really appreciate your content. Please let me know.
Thanks
buy viagra online
Hello there! I could have sworn I’ve been to this site before but after browsing through
a few of the articles I realized it’s new to me.
Nonetheless, I’m definitely pleased I came across it and I’ll be bookmarking it and checking back frequently!
klksymjau hvbnt zgbuiwm rhrm eoumdfnldecvemd
buy viagra online
buy viagra online
watch porn video
buy viagra online
payday loan
payday loan
payday loan
payday loan
buy viagra online
payday loan
payday loan
I really like what you guys tend to be up too.
This sort of clever work and reporting! Keep up the superb works guys I’ve added you guys to blogroll.
buy viagra online
watch porn video
payday loan
bookmarked!!, I really like your site!
buy viagra online
Hi there to every , for the reason that I am really keen of reading
this web site’s post to be updated daily. It carries good material.
buy viagra online
buy viagra online
My brother suggested I might like this website. He was entirely
right. This post truly made my day. You cann’t imagine simply how much time I had spent for this information! Thanks!
Do you mind if I quote a few of your articles as long as I provide credit and sources back to your weblog?
My blog is in the exact same area of interest as yours and my users would genuinely benefit
from some of the information you provide here.
Please let me know if this ok with you. Thank you!
payday loan
My brother recommended I might like this website. He was entirely right.
This post actually made my day. You cann’t imagine simply how much time I had
spent for this information! Thanks!
buy viagra online
Keep on writing, great job!
penis enlargement
buy viagra online
watch porn video
It’s amazing to go to see this website and reading the views of all colleagues concerning this article,
while I am also eager of getting experience.
What’s up colleagues, fastidious post and good urging commented here, I am in fact enjoying by these.
Wow, this post is good, my younger sister is analyzing these things,
so I am going to tell her.
my blog; calendrier mai
buy viagra online
payday loan
penis enlargement
payday loan
penis enlargement
watch porn video
penis enlargement
buy viagra online
penis enlargement
I was wondering if you ever considered changing the layout of your blog?
Its very well written; I love what youve got to say.
But maybe you could a little more in the way of content so
people could connect with it better. Youve got an awful lot of text for only
having 1 or 2 images. Maybe you could space it out better?
Look into my website … printable blank monthly calendar
payday loan
buy viagra online
watch porn video
buy viagra online
buy viagra online
Hey! This is my first visit to your blog! We are a collection of volunteers and starting a new
project in a community in the same niche. Your blog provided us useful information to work on. You have done a wonderful
job!
payday loan
Appreciation to my father who shared with me concerning this website, this webpage is really amazing.
buy viagra online
buy viagra online
payday loan
buy viagra online
watch porn video
buy viagra online
penis enlargement
payday loan
buy viagra online
What i don’t understood is actually how you’re no longer actually a
lot more neatly-favored than you may be right now.
You’re very intelligent. You already know thus considerably when it comes
to this subject, made me in my opinion imagine it from numerous
numerous angles. Its like men and women are not fascinated unless it is something to accomplish with Girl gaga!
Your personal stuffs great. At all times handle it up!
My page; Newest cars coloring pages
penis enlargement
payday loan
watch porn video
buy viagra online
If you are going for most excellent contents like me, only go
to see this site all the time for the reason that it gives
feature contents, thanks
buy viagra online
watch porn video
Hi are using WordPress for your blog platform? I’m new to
the blog world but I’m trying to get started and create my own. Do you need any html coding expertise
to make your own blog? Any help would be really appreciated!
Online poker
I simply could not go away your web site prior to suggesting that I extremely loved the usual info an individual provide in your guests? Is going to be back incessantly in order to inspect new posts.
buy viagra online
buy viagra online
When someone writes an post he/she keeps the
plan of a user in his/her brain that how a user can know it.
Thus that’s why this post is outstdanding.
Thanks!
payday loan
buy viagra online
buy viagra online
penis enlargement
buy viagra online
penis enlargement
payday loan
payday loan
buy viagra online
It’s very simple to find out any matter on web as compared to books, as I found this article at this website.
Feel free to surf to my site paperblanks kalender 2023
penis enlargement
buy viagra online
Este site é realmente demais. Sempre que acesso eu encontro coisas boas Você também vai querer acessar o nosso site e saber mais detalhes! Conteúdo exclusivo. Venha saber mais agora! 🙂
buy viagra online
Thanks for sharing superb informations. Your site is very cool. I’m impressed by the details that you have on this web site. It reveals how nicely you understand this subject. Bookmarked this web page, will come back for extra articles. You, my friend, ROCK! I found just the info I already searched all over the place and just couldn’t come across. What a perfect web site.
penis enlargement
This is really interesting, You’re a very skilled blogger.
I’ve joined your rss feed and look forward to seeking more of your
magnificent post. Also, I’ve shared your web site in my social networks!
Hey there superb blog! Does running a blog similar to
this require a great deal of work? I have very little
understanding of coding but I had been hoping to start my own blog in the near future.
Anyways, should you have any recommendations or techniques for new blog owners please share.
I understand this is off subject however I just needed to ask.
Thank you!
Hello i am kavin, its my first time to commenting anywhere, when i
read this paragraph i thought i could also make comment due to this good post.
I’ve been exploring for a little for any high quality articles or blog posts in this kind of area .
Exploring in Yahoo I ultimately stumbled upon this site.
Studying this information So i’m happy to exhibit that I’ve an incredibly just right uncanny feeling I discovered exactly what I needed.
I such a lot indubitably will make certain to do not fail to remember
this site and provides it a glance regularly.
{Кредит онлайн}
Fabulous, what a website it is! This weblog gives useful information to us,
keep it up.
I’m curious to find out what blog platform you have been working with?
I’m having some minor security issues with my latest website and I’d like to find something more safe.
Do you have any solutions?
We’re a bunch of volunteers and opening a new scheme in our community.
Your website offered us with valuable info to work on. You have performed an impressive process and our entire community might be grateful to you.
What i don’t realize is in fact how you are no longer actually much
more smartly-appreciated than you might be right now. You are so intelligent.
You understand thus significantly relating to
this matter, made me personally believe it from a lot of numerous
angles. Its like men and women aren’t fascinated unless it’s one
thing to accomplish with Lady gaga! Your personal stuffs great.
All the time handle it up![url=https://backlinkonlyy.blogspot.com/]เพิ่ม แรงม้า[/url]
เพิ่ม แรงม้า
Tremendous issues here. I’m very satisfied to look your post.
Thank you a lot and I’m looking ahead to touch you.
Will you please drop me a e-mail?
Download free anime wallpapers
Simply want to say your article is as amazing. The clarity
to your submit is simply great and that i could assume you are knowledgeable on this subject.
Fine along with your permission let me to seize your feed to keep up to date with drawing close post.
Thank you a million and please keep up the
rewarding work.
Admiring the time and energy you put into your blog and detailed
information you provide. It’s great to come across a blog every once in a while that isn’t the same
out of date rehashed material. Wonderful read! I’ve saved your site and I’m
including your RSS feeds to my Google account.
Definitely believe that which you said. Your favorite justification seemed to be on the web
the simplest thing to be aware of. I say to you, I definitely get
annoyed while people consider worries that they plainly
don’t know about. You managed to hit the nail upon the top and also defined out the whole thing without having side-effects
, people could take a signal. Will likely be back to get
more. Thanks
URL
Keywords سمساری در ونک
Blog_Comment دفتر ازدواج شمال تهران با
معتبریت خود، تضمین کننده پذیرایی با شامی شگفتانگیز، سفره عقدی
با گلهای طبیعی و دیزاین زیبا است.
Anchor_Text سمساری در ونک
Image_Comment بسیاری از این لوازم لوکس هستند
و یا لوازمی هستند که در زندگی روزمره
مورد استفاده قرار می گیرند.
Guestbook_Comment آنها وضعیت محصول را بررسی کرده و قیمت پیشنهادی را به
شما اعلام میکنند.
Category misc
Micro_Message مورد دیگر، قیمت متفاوت
این لوازم می باشد که خرید آنها را برای
عموم دلچسب میکند.
About_Yourself 46 year-old Nuclear Power Engineer Doretta Hegarty, hailing from Bow Island enjoys watching movies like Vehicle 19
and Board sports. Took a trip to Historic City of Sucre and drives a
Ferrari 250 GT SWB California Spyder.
Forum_Comment سمساری آنلاین در دستههای مختلفی از محصولات و خدمات
مانند الکترونیک، لوازم خانگی، خودرو، ملک، لوازم شخصی، خدمات مالی و غیره قابل
انجام است.
Forum_Subject ️سمساری در دبستان تهران سمساری 09122725127 سمسارچی ️
Video_Title سمساری در ونک 09363333948 سمساری برتر تهران
Video_Description اکثر ساخت و سازهای این محله پس از انقلاب صورت گرفت و امروزه شاهد طراحی و شهرسازی
متفاوتی در این محله هستید.
Preview_Image
YouTubeID
Website_title ️سمساری در نعمت آباد تهران سمساری 09122725127 سمسارچی ️
Description_250 سمساری سمسارچی یکی از بهترین سمساری ها
در دبستان تهران می باشد.
Guestbook_Comment_(German)
Description_450 برای فروختن کالا های دست دوم، رو هایی مختلفی وجود دارد اما یکی از
معتبرترین آنها فروختن این اجناس به سمساری هاست.
Guestbook_Title لیست آدرس ، تلفن سمساری نزدیک , محله
ونک شمال و شرق تهران منطقه 3, شهر اینترنتی تهران
Website_title_(German)
Description_450_(German)
Description_250_(German)
Guestbook_Title_(German)
Image_Subject سمساری سمساری آفتاب سمساری تهران 09120907647
Website_title_(Polish)
Description_450_(Polish)
Description_250_(Polish)
Blog Title سمساری آنلاین تهران ونک،
تهران نقشه نشان
Blog Description ️سمساری در آریاشهر سمساری 09122725127 سمسارچی ️
Company_Name سمساری در ونک
Blog_Name لیست آدرس ، تلفن سمساری نزدیک , محله ونک شمال و شرق تهران منطقه
3, شهر اینترنتی تهران
Blog_Tagline ️فروش ساید بای ساید دست
دوم در تهران سمساری 09122725127
سمسارچی ️
Blog_About 31 yrs old VP Accounting Glennie Pullin, hailing
from Windsor enjoys watching movies like Downhill and Vehicle restoration. Took a
trip to Cidade Velha and drives a Ferrari 512.
Article_title خریدار فوری لوازم منزل به قیمت روز سمساری در تمام مناطق
Article_summary با سمسارچی، خریدار انواع کولر گازی در تهران –
خریدار انواع کولر آبی در تهران در تماس باشید.
Article وجه نامگذاری آن به علت فراوانی گل
پونه در دره پونک در قدیم است. قدمت این ده به بیش از هشتصد سال میرسد و درخت بید معروفی در محله وجود دارد که نشاندهنده این قدمت است.
ده پونک در قدیم محل باغهای بزرگ و توتستانهای قدیمی غرب تهران بودهاست که از این باغها شمار
کمی امروزه بهجا ماندهاست.
نزدیکترین ایستگاه تاکسیرانی به محله پونک از جمله ایستگاه میدان پونک به میدان ولیعصر،
میدان ونک به میدان صنعت و سیدخندان
است.
About_Me 25 year-old Community Outreach Specialist Teador Murphy, hailing from Sainte-Genevieve enjoys watching movies like Colonel Redl (Oberst Redl) and
Knapping. Took a trip to Cidade Velha and drives
a Ferrari 250 GT LWB California Spider.
About_Bookmark 39 years old Developer II Alessandro Fishpoole,
hailing from Angus enjoys watching movies like “Corrina, Corrina” and Mushroom hunting.
Took a trip to Works of Antoni Gaudí and drives a Ferrari 375
MM Berlinetta.
Topic سمساری در ونک
I was suggested this web site via my cousin. I’m not certain whether or not this submit is
written by means of him as no one else recognise such exact approximately my problem.
You’re wonderful! Thanks!
Everything is very open with a really clear clarification of the challenges.
It was definitely informative. Your website is useful. Thank you for sharing!
An outstanding share! I’ve just forwarded this onto a colleague
who has been conducting a little homework on this.
And he in fact ordered me dinner because I discovered it for him…
lol. So let me reword this…. Thanks for the meal!!
But yeah, thanks for spending the time to talk about this matter here on your website.
demais este conteúdo. Gostei muito. Aproveitem e vejam este site. informações, novidades e muito mais. Não deixem de acessar para aprender mais. Obrigado a todos e até mais. 🙂
I’m not sure why but this website is loading very slow for me.
Is anyone else having this problem or is it a problem on my end?
I’ll check back later and see if the problem still exists.
Hi! I realize this is sort of off-topic however I needed to ask.
Does building a well-established blog such aas yours take a massive amount work?
I am brand new to operating a blog however I do write in my journal daily.
I’d like to start a blog so I can easily share my own experience and
views online. Please let me know if you have any ideas or tips
for brand new aspiring bloggers. Appreciate it!
web site
Youu additionallly need to compliment it with a quick working pc so that it can handle the
processes of all your browsing and enjoying. Gett the newest on new casino developments, economics, laws and more.
As such, too seek out the best online on line casino site for
you, then you definately want to consider what finances
you will play with.
This post presents clear idea for the new viewers of blogging, that actually how to do running a blog.
Incredible! This blog looks just like my old one! It’s on a totally different subject but it has pretty much
the same layout and design. Great choice of colors!
What’s up mates, nice article and fastidious arguments commented
here, I am actually enjoying by these.
This is very interesting, You are a very skilled blogger.
I have joined your rss feed and look forward to seeking
more of your excellent post. Also, I have shared your web site in my social
networks!
Thanks in favor of sharing such a good thinking, article is pleasant, thats why i have read it completely
Hi! I know this is kinda off topic but I’d figured I’d
ask. Would you be interested in trading links or maybe guest authoring a blog article or vice-versa?
My site discusses a lot of the same topics as yours and I believe
we could greatly benefit from each other. If you’re interested feel free
to shoot me an email. I look forward to hearing from you!
Awesome blog by the way!
I’m gone to inform my little brother, that
he should also go to see this webpage on regular basis to get updated from newest news update.
It’s hard to come by well-informed people in this particular topic, however, you sound like you know what you’re talking about!
Thanks
It’s a pity you don’t have a donate button! I’d definitely
donate to this brilliant blog! I guess for now i’ll settle for bookmarking and adding your RSS feed to my Google account.
I look forward to new updates and will talk about this site with my Facebook group.
Chat soon!
Have you ever thought about including a little bit more than just your articles?
I mean, what you say is fundamental and everything.
But just imagine if you added some great pictures or videos to give your posts more, “pop”!
Your content is excellent but with pics and video clips, this website could certainly be one of the most beneficial in its niche.
Good blog!
Have you ever thought about creating an e-book or guest authoring
on other websites? I have a blog based upon on the same
subjects you discuss and would really like to have you share some stories/information. I know my viewers would enjoy your work.
If you’re even remotely interested, feel free to send me an email.
Please let me know if you’re looking for a writer for your weblog.
You have some really great articles and I feel I would be a good asset.
If you ever want to take some of the load off, I’d absolutely love to write some material for your blog in exchange for a link back to mine.
Please blast me an email if interested. Regards!
I know this website offers quality depending posts and other
data, is there any other web page which provides these kinds of information in quality?
Heya i am for the primary time here. I came across this board and I in finding It really helpful & it helped me
out much. I hope to give something again and help
others such as you helped me.
Pretty! This has been an extremely wonderful
article. Thanks for providing this information.
I am extremely inspired with your writing abilities and also with the
format in your weblog. Is that this a paid subject matter or did you customize it your
self? Either way keep up the excellent quality writing, it is rare to see a nice blog like this one these days..
My developer is trying to convince me to move to .net from
PHP. I have always disliked the idea because of the expenses.
But he’s tryiong none the less. I’ve been using WordPress on a variety of websites for about a year and am anxious about switching to another platform.
I have heard excellent things about blogengine.net.
Is there a way I can import all my wordpress posts into it?
Any help would be greatly appreciated!
Including disrespect, dishonesty, controlling actions, or a absence of support.
In a healthy partnership, everything type associated with functions just. Sure, you may disagree from period to period or arrive upon additional bumps in the street, but you usually create choices jointly, discuss any complications that arise openly, and truly appreciate each additional’t business.
penis enlargement
Online poker
Este site é realmente fantástico. Sempre que acesso eu encontro coisas boas Você também pode acessar o nosso site e descobrir detalhes! informaçõesexclusivas. Venha descobrir mais agora! 🙂
I’m gone to convey my little brother, that he should also pay a quick
visit this web site on regular basis to take updated from newest information.
Merely a smiling visitant here to share the love (:, btw outstanding style. “Individuals may form communities, but it is institutions alone that can create a nation.” by Benjamin Disraeli.
Hello to every , for the reason that I am really eager of reading this web site’s post
to be updated on a regular basis. It carries nice information.
It’s really a great and useful piece of information. I am glad that you shared this helpful information with us. Please keep us up to date like this. Thanks for sharing.
I know this web page gives quality based posts and additional data,
is there any other web site which presents such data in quality?
Bocoran pasaran Sydney terbaru
buy viagra online
I think this is among the most vital info for me.
And i am glad reading your article. But want to remark on some general
things, The web site style is wonderful, the articles
is really great : D. Good job, cheers
Here is my web site: Tratamentos
Nice blog here! Also your website loads up fast! What web host are
you using? Can I get your affiliate link to your host?
I wish my site loaded up as fast as yours lol
I all the time used to read post in news papers but now as I am a
user of net thus from now I am using net for articles or reviews, thanks
to web.
I must thank you for the efforts you’ve put in writing this website.
I really hope to view the same high-grade blog posts from you later on as well.
In fact, your creative writing abilities has motivated me to get my own website
now 😉
Hi there, this weekend is good designed for me, since this point in time i am reading
this impressive educational post here at my house.
Superb blog! Do you have any helpful hints for aspiring writers?
I’m hoping to start my own blog soon but I’m a little lost on everything.
Would you advise starting with a free platform
like WordPress or go for a paid option? There are so many options out there that
I’m completely overwhelmed .. Any recommendations?
Cheers!
Link exchange is nothing else except it is only placing the other person’s webpage link on your page at suitable place and other person will also do same for you.
watch porn video
If some one desires to be updated with newest technologies then he must be pay a quick visit this web site and be up
to date everyday.
Hi there, this weekend is nice designed for me, since this moment
i am reading this enormous informative post here at my
home.
Simply want to say your article is as amazing. The clearness on your
submit is just nice and i can suppose you are an expert in this subject.
Well with your permission let me to grab your
RSS feed to stay up to date with approaching post. Thank you a
million and please carry on the gratifying work.
Hello there, There’s no doubt that your site could be having internet browser compatibility issues.
When I take a look at your website in Safari, it looks fine
however, if opening in Internet Explorer, it’s got some overlapping
issues. I merely wanted to provide you with a quick heads
up! Aside from that, fantastic website!
When someone writes an article he/she maintains the
thought of a user in his/her brain that how
a user can understand it. Therefore that’s why this
post is outstdanding. Thanks!
I am now not certain the place you are getting your information,
however good topic. I must spend some time studying more or understanding more.
Thank you for magnificent information I was looking for this information for my mission.
Thanks for sharing your thoughts on 정품비아그라.
Regards
I’m not sure where you’re getting your info, but great topic.
I needs to spend some time learning much more or understanding more.
Thanks for fantastic info I was looking for this info for
my mission.
Do you have any video of that? I’d love to find out more details.
Heya i’m for the first time here. I came across this board and I find It really useful & it helped me out
a lot. I hope to give something back and help others like you aided me.
buy viagra online
After looking over a few of the blog posts on your blog, I honestly like your technique of writing a blog.
I book marked it to my bookmark website list and will be checking
back soon. Please check out my website as well and tell me what you think.
Adorei este site. Pra saber mais detalhes acesse nosso site e descubra mais. Todas as informações contidas são informações relevantes e únicos. Tudo que você precisa saber está ta lá.
สล็อต เว็บใหญ่ อันดับ
1,เว็บใหญ่สล็อต,เว็บ ใหญ่ สล็อต,เกมสล็อตเว็บใหญ่,สล็อต เว็บ ใหญ่ ที่สุด pg,สล็อต เว็บ
ใหญ่ อันดับ 1,เกมสล็อตอันดับ 1,สล็อต เว็บใหญ่,เว็บสล็อตใหญ่ที่สุด,สล็อตเว็บใหญ่ pg,เว็บสล็อต
ที่ มี คน เล่น มาก ที่สุด,สล็อตเว็บใหญ่ที่สุดในโลก,เว็บ สล็อต ใหญ่
ๆ,สล็อต เว็บ ใหญ่ เว็บ ตรง,สล็อตเว็บใหญ่ที่สุด
It’s in fact very difficult in this active life to listen news on Television, therefore I only use world wide
web for that reason, and take the newest news.
Simply desire to say your article is as astonishing. The clearness to
your publish is just nice and i could assume you’re an expert in this subject.
Well along with your permission allow me to grab your feed
to stay updated with drawing close post. Thank you a million and please keep up the gratifying work.
Ebony huge butts have lots of various titles- fanny, butt, buns, and booty.https://www.visualpixel.es/2016/06/13/aliquam-erat-volutpat-2/ Fanny will be a phrase that’s even more mainstream and much less intimate, therefore anyone can use it. Bottom is certainly usually utilized in a jokey way. Buns sound more attractive, and behind is noticed as kinky. In Traditional western culture, the dimension of a woman’s booty, boobs, and waist size determines how desirable she is usually. Caucasian females tend to prefer bigger boobs and a regular dimension butt, as lengthy as it’s not really level. But ebony women are usually proud of their big, juicy butts and enjoy to show them off. It’s something they make use of to their benefit, and porno fans all over the global entire world understand this. Just look at J. Lo – she may be a great dancer and vocalist, but it’s her well-known behind that put her in the spotlight. It’s even rumored to become covered!
Howdy! I know this is kinda off topic but I was wondering if you knew where I could locate a captcha plugin for my comment form?
I’m using the same blog platform as yours and I’m having problems finding one?
Thanks a lot!
My brother suggested I may like this website.
He was entirely right. This publish actually made my day.
You can not believe just how a lot time I had spent for this information! Thanks!
I every time spent my half an hour to read this weblog’s articles or reviews all the time along with
a mug of coffee.[url=https://penzu.com/public/d77c13c0cb1a8a9c]รีแมพดีไหม[/url]
รีแมพคืออะไร
Este site é realmente fabuloso. Sempre que consigo acessar eu encontro novidades Você também vai querer acessar o nosso site e saber mais detalhes! informaçõesexclusivas. Venha saber mais agora! 🙂
Undeniably believe that which you stated. Your favorite justification appeared to be
on the web the simplest thing to be aware of. I say to you, I definitely get irked while people think about worries that they just do not know
about. You managed to hit the nail upon the top as well as defined out
the whole thing without having side effect , people could take a signal.
Will probably be back to get more. Thanks
Wonderful blog! Do you have any helpful hints for aspiring writers?
I’m hoping to start my own website soon but I’m a little lost on everything.
Would you propose starting with a free platform like WordPress or go
for a paid option? There are so many choices out there that I’m totally confused ..
Any ideas? Thank you!
Do you mind if I quote a couple of your posts as long as I provide
credit and sources back to your webpage? My blog is in the very
same area of interest as yours and my visitors would really benefit from
some of the information you provide here. Please let
me know if this okay with you. Thanks!
Can you tell us more about this? I’d care to find out some additional information.
I used to be recommended this blog by means of my cousin. I am now
not positive whether this put up is written through him as
no one else understand such specified about my trouble.
You’re wonderful! Thank you!
I am not sure where you’re getting your info, but great topic.
I needs to spend some time learning more or understanding more.
Thanks for wonderful information I was looking for this information for my mission.
Magnificent goods from you, man. I’ve understand your stuff previous to and you
are just extremely magnificent. I really like what you’ve acquired here, really like what you are saying
and the way in which you say it. You make it enjoyable
and you still take care of to keep it sensible. I cant wait to read much more from you.
This is really a terrific web site.
I love reading through a post that will make people think.
Also, thanks for permitting me to comment!
Hi there to all, because I am truly keen of reading this blog’s post
to be updated on a regular basis. It consists of good stuff.
Heya just wanted to give you a quick heads up and let
you know a few of the images aren’t loading correctly. I’m
not sure why but I think its a linking issue.
I’ve tried it in two different web browsers and both show the same outcome.
I do trust all the ideas you have presented in your post.
They’re really convincing and will definitely work.
Still, the posts are very short for novices. May you please extend them a little from next time?
Thanks for the post.
Excellent blog here! Also your site loads up fast! What web host are you using?
Can I get your affiliate link to your host? I wish my site loaded up
as fast as yours lol
You actually make it appear really easy together with your presentation but
I to find this topic to be really something that I think I might by no means understand.
It kind of feels too complicated and extremely wide for me.
I’m having a look ahead on your next submit, I’ll try to get
the hang of it!
Right now it looks like BlogEngine is the top blogging platform out there
right now. (from what I’ve read) Is that what you are using on your blog?
Excellent weblog right here! Also your site lots up very fast!
What host are you the usage of? Can I get your affiliate link
in your host? I wish my site loaded up as fast as yours lol
Wonderful article! We will be linking to this great article on our site.
Keep up the great writing.
Wow, awesome weblog format! How lengthy have you been blogging for?
you make blogging glance easy. The total glance of your website is fantastic, as
well as the content!
Excellent, what a website it is! This web site gives
helpful information to us, keep it up.
This page really has all of the information I needed about this subject
and didn’t know who to ask.
I was suggested this website by my cousin. I am not sure whether
this post is written by him as nobody else know such detailed about my trouble.
You’re wonderful! Thanks!
Fantastic beat ! I wish to apprentice while you amend your website, how could i subscribe
for a blog website? The account helped me a acceptable deal.
I had been tiny bit acquainted of this your broadcast offered bright clear concept
Greetings! Very helpful advice in this particular post!
It’s the little changes which will make the largest changes.
Thanks a lot for sharing!
I like the helpful information you provide in your articles.
I’ll bookmark your weblog and test again here
frequently. I’m somewhat certain I will be told plenty of new stuff right here!
Best of luck for the next!
We are a group of volunteers and starting a new
scheme in our community. Your web site offered us with valuable info to work on. You’ve done a formidable job and our whole
community will be grateful to you.
Este site é realmente demais. Sempre que acesso eu encontro coisas boas Você também pode acessar o nosso site e saber mais detalhes! conteúdo único. Venha saber mais agora! 🙂
Hello! I could have sworn I’ve been to this blog before but after browsing through some of the post I realized it’s new to me. Anyways, I’m definitely happy I found it and I’ll be book-marking and checking back frequently!